Neurocrine Biosciences Announces Publication of Expert Recommendations for Glucocorticoid Dose Reduction after Initiating CRENESSITY® (crinecerfont) for the Treatment of Classic Congenital Adrenal Hyperplasia
PR Newswire
SAN DIEGO, May 6, 2026
- New algorithms developed by expert clinical endocrinologists provide clear, practical recommendations for healthcare providers to reduce supraphysiologic glucocorticoid (GC) doses in patients with classic congenital adrenal hyperplasia (CAH) after starting CRENESSITY
- CRENESSITY reduces excess androgen production, enabling patients with classic CAH to move toward more physiologic GC dosing while maintaining essential daily cortisol replacement
SAN DIEGO, May 6, 2026 /PRNewswire/ — Neurocrine Biosciences, Inc. (Nasdaq: NBIX) today announced the publication of the first peer-reviewed expert recommendations to guide glucocorticoid dose reduction in patients with classic congenital adrenal hyperplasia treated with CRENESSITY® (crinecerfont). Published in The Journal of Clinical Endocrinology & Metabolism, the recommendations address a critical unmet need as real-world use of CRENESSITY expands in both pediatric and adult patients.
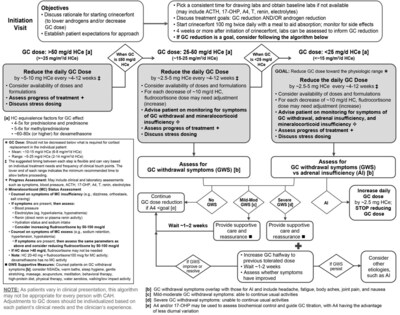
The recommendations for clinicians treating classic congenital adrenal hyperplasia (CAH) patients with CRENESSITY are presented in two complementary manuscripts — one focused on pediatric patients aged four to 17 years and one focused on adults. The publications include the first structured, expert-driven algorithms for reducing supraphysiologic glucocorticoid (GC) dosing after initiating CRENESSITY in real-world clinical practice. CRENESSITY is approved as an adjunct treatment to GC replacement to control androgens in adult and pediatric patients four years of age and older with classic CAH.
“By reducing ACTH and downstream androgen production, CRENESSITY allows patients with classic CAH, including both salt-wasting and simple virilizing forms, to move toward more physiologic, cortisol-replacing glucocorticoid dosing,” said Sanjay Keswani, M.D., Chief Medical Officer, Neurocrine Biosciences. “Blocking excess ACTH and androgen production while maintaining necessary cortisol replacement reflects an important shift in how the disease is managed, helping to lower risks associated with long-term exposure to supraphysiologic glucocorticoid doses.”
In addition to the protocols used in the CAHtalyst® Pediatric and Adult Phase 3 clinical trials, clinicians now have expert-developed systematic approaches to adjusting GC doses in pediatric and adult patients treated with CRENESSITY. These recommendations, grounded in both clinical trial and real-world clinical experience, recognize that treatment goals and clinical considerations differ meaningfully between children and adults and provide two distinct algorithms tailored to each population:
- Pediatric patients: The expert recommendations center on support to achieve normal growth and normalize bone age maturation and pubertal development while reducing long-term complications of excess GCs. GC reductions are targeted toward the upper portion of the physiologic range (8-11 mg/m2/day in hydrocortisone equivalents; full range 4-11 mg/m2/day) and are guided primarily by androgen concentrations, with close monitoring for GC withdrawal symptoms and adequate cortisol and mineralocorticoid replacement.
- Adult patients: The recommendations focus on minimizing GC-related metabolic, cardiovascular and skeletal complications while maintaining androgen control. GC reductions are targeted toward the physiologic range of 2-14 mg/m2/day in hydrocortisone equivalents and are paced according to current dose to manage any GC withdrawal symptoms and monitor for adequate cortisol replacement.
“In children and adolescents with classic congenital adrenal hyperplasia, treatment decisions must carefully balance disease control with normal growth and development,” said Mimi Kim, M.D., M.Sc., Associate Professor of Clinical Pediatrics, Keck School of Medicine, University of Southern California. “These recommendations provide clinicians with a structured, practical approach to glucocorticoid management that helps support growth, bone maturation and pubertal development as CRENESSITY is incorporated into care.”
“CRENESSITY enables us to decouple androgen control from the need for supraphysiologic glucocorticoids in patients with classic congenital adrenal hyperplasia,” said Oksana Hamidi, D.O., M.S.C.S., Associate Professor of Internal Medicine, Division of Endocrinology, UT Southwestern Medical Center. “This new framework provides an algorithm and practical steps clinicians can take to lower glucocorticoid exposure without compromising cortisol replacement, helping to realize the benefits of this novel therapeutic approach in day-to-day care.”
GC reduction can offer important benefits for many patients and is therefore a common treatment goal with CRENESSITY. However, for some patients already taking a physiologic GC dose, CRENESSITY may be initiated to better manage androgens without increasing the GC dose. Key considerations outlined in both manuscripts include:
- Dose reductions should be gradual and clinically supervised, with ongoing assessment for symptoms of GC withdrawal, adrenal insufficiency and mineralocorticoid imbalance.
- Healthcare providers should consider monitoring key biomarkers, which may include androstenedione, 17-hydroxyprogesterone, adrenocorticotropic hormone (ACTH), testosterone and markers of mineralocorticoid status, such as renin and electrolytes.
- GC dose reduction is not a universal goal; for some patients, the focus is androgens. The expert recommendation highlights that some individuals may already be receiving a physiologic GC dose and may initiate treatment with CRENESSITY solely for androgen control.
- Dose reduction should be considered only when appropriate, including when androgens are at or below goal and when clinical goals support reducing supraphysiologic GC exposure.
The publications and new clinical algorithms can be accessed here:
1 Glucocorticoid Reduction After Starting Crinecerfont in Pediatric Patients with Classic CAH: Practical Perspectives.
2 Glucocorticoid Reduction After Starting Crinecerfont in Adult Patients with Classic CAH: Practical Perspectives.
*Figure reproduced from Nokoff NJ et al. J Clin Endocrinol Metab; 2026,1 distributed under the terms of the Creative Commons CC BY license
†Figure reproduced from Hamidi O, et al. J Clin Endocrinol Metab. 2026,2 distributed under the terms of the Creative Commons CC BY license
About Congenital Adrenal Hyperplasia
Congenital adrenal hyperplasia (CAH) is a rare genetic condition that results in an enzyme deficiency that alters the production of adrenal steroid hormones, such as cortisol, aldosterone and adrenal androgens. Severe enzyme deficiency leads to an inability of the adrenal glands to produce enough cortisol and, in approximately 75% of cases, aldosterone. Because individuals with CAH are typically still able to produce androgens, the unused precursors that would normally be used to make cortisol instead result in the production of excess amounts of androgens. If left untreated, CAH can result in adrenal crisis and even death.
Exogenous glucocorticoids (GCs) are necessary to correct the endogenous cortisol deficiency, but historically, doses higher than those needed for cortisol replacement (supraphysiologic) have been used to lower the elevated levels of adrenocorticotropic hormone (ACTH) and adrenal androgens. However, GC treatment at supraphysiologic doses has been associated with serious and significant complications of steroid excess, including metabolic issues such as weight gain and diabetes, cardiovascular disease and osteoporosis. Additionally, long-term treatment with supraphysiologic GCs may have psychological and cognitive impacts, such as changes in mood and memory. Adrenal androgen excess has been associated with abnormal bone growth and development in pediatric patients, female health problems such as excess facial hair growth and menstrual irregularities, in addition to cardiometabolic and fertility issues in both sexes. The symptoms of high ACTH may include testicular adrenal rest tumors (TARTs).
About CRENESSITY® (crinecerfont)
CRENESSITY is a potent and selective oral corticotropin-releasing factor type 1 receptor (CRF1) antagonist that reduces and controls excess adrenocorticotropic hormone (ACTH) and adrenal androgens through a non-glucocorticoid (GC) mechanism for the treatment of classic congenital adrenal hyperplasia (CAH). Antagonism of CRF1 receptors in the pituitary has been shown to decrease ACTH levels, which in turn decreases the production of adrenal androgens and potentially the symptoms associated with CAH. The robust clinical study data demonstrate that lowering adrenal androgen levels with CRENESSITY enables lower, more physiologic dosing of GCs to replace missing cortisol.
CRENESSITY comes in capsules and an oral solution. For adults 18 years of age and older, the recommended dosage is 100 mg twice daily taken orally with a meal. For pediatric patients four to 17 years of age weighing less than 55 kg (121 lbs), the recommended dosage is based on body weight and is administered twice daily, taken orally with a meal. For pediatric patients weighing more than 55 kg (121 lbs), the recommended dosage is 100 mg twice daily taken orally with a meal. Healthcare providers can work with patients to determine the appropriate formulation for use depending on patient needs. Patients receiving CRENESSITY should continue GC therapy for cortisol replacement.
About The CAHtalyst® Studies
The Phase 3 CAHtalyst global registrational studies were designed to evaluate the safety, efficacy and tolerability of CRENESSITY® (crinecerfont) in children and adults with classic congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency. The CAHtalyst studies were the largest-ever clinical trial program in classic CAH, including 285 pediatric and adult patients.
The CAHtalyst Pediatric study included 103 pediatric patients four to 17 years of age. The study tested two questions. The first question evaluated whether four weeks of CRENESSITY treatment could improve androgen control. The second question evaluated whether an additional 24 weeks of CRENESSITY treatment enabled customized glucocorticoid (GC) down-titration while androstenedione levels were maintained or improved.
The CAHtalyst Adult study included 182 adult patients 18 to 58 years of age. Similarly, the first question of the study evaluated whether four weeks of CRENESSITY treatment could improve androgen control, and the second question evaluated whether an additional 20 weeks of CRENESSITY treatment enabled GC reduction to physiologic range while androstenedione levels were maintained or improved.
Data from the CAHtalyst Phase 3 studies supported approval of CRENESSITY by the U.S. Food and Drug Administration in December 2024. The open-label extension treatment portions of both studies are ongoing.
Important Information
Approved Uses
CRENESSITY® (crinecerfont) is a prescription medicine used together with glucocorticoids (steroids) to control androgen (testosterone-like hormone) levels in adults and children 4 years of age and older with classic congenital adrenal hyperplasia (CAH).
IMPORTANT SAFETY INFORMATION
Do not take CRENESSITY if you:
Are allergic to crinecerfont, or any of the ingredients in CRENESSITY.
CRENESSITY may cause serious side effects, including:
Allergic reactions. Symptoms of an allergic reaction include tightness of the throat, trouble breathing or swallowing, swelling of the lips, tongue, or face, and rash. If you have an allergic reaction to CRENESSITY, get emergency medical help right away and stop taking CRENESSITY.
Risk of Sudden Adrenal Insufficiency or Adrenal Crisis with Too Little Glucocorticoid (Steroid) Medicine. Sudden adrenal insufficiency or adrenal crisis can happen in people with congenital adrenal hyperplasia who are not taking enough glucocorticoid (steroid) medicine. You should continue taking your glucocorticoid (steroid) medicine during treatment with CRENESSITY. Certain conditions such as infection, severe injury, or shock may increase your risk for sudden adrenal insufficiency or adrenal crisis. Tell your healthcare provider if you get a severe injury, infection, illness, or have planned surgery during treatment. Your healthcare provider may need to change your dose of glucocorticoid (steroid) medicine.
Before taking CRENESSITY, tell your healthcare provider about all of your medical conditions, including if you: are pregnant or plan to become pregnant, or are breastfeeding or plan to breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements.
The most common side effects of CRENESSITY in adults include tiredness, headache, dizziness, joint pain, back pain, decreased appetite, and muscle pain.
The most common side effects of CRENESSITY in children include headache, stomach pain, tiredness, nasal congestion, and nosebleeds.
These are not all the possible side effects of CRENESSITY. Call your healthcare provider for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit MedWatch at www.fda.gov/medwatch or call 1-800-FDA-1088.
Dosage Forms and Strengths: CRENESSITY is available in 50 mg and 100 mg capsules, and as an oral solution of 50 mg/mL.
Please see full Prescribing Information.
About Neurocrine Biosciences, Inc.
Neurocrine Biosciences is a leading biopharmaceutical company with a simple purpose: to relieve suffering for people with great needs. We are dedicated to discovering and developing life-changing treatments for patients with under-addressed neurological, endocrine, psychiatric and immunological disorders. The company’s diverse portfolio includes FDA-approved treatments for tardive dyskinesia, chorea associated with Huntington’s disease, classic congenital adrenal hyperplasia, endometriosis* and uterine fibroids*, as well as a robust pipeline including multiple compounds in mid- to late-phase clinical development across our core therapeutic areas. For three decades, we have applied our unique insight into neuroscience and the interconnections between brain and body systems to treat complex conditions. We relentlessly pursue medicines to ease the burden of debilitating diseases and disorders because you deserve brave science. For more information, visit neurocrine.com, and follow the company on LinkedIn, X, Facebook and YouTube. (*in collaboration with AbbVie)
The NEUROCRINE BIOSCIENCES Logo, NEUROCRINE, YOU DESERVE BRAVE SCIENCE, CRENESSITY and CAHtalyst are registered trademarks of Neurocrine Biosciences, Inc.
Forward-Looking Statements
In addition to historical facts, this press release contains forward-looking statements that involve a number of risks and uncertainties. These statements include, but are not limited to, statements regarding the potential benefits to be derived from CRENESSITY for the treatment of classic congenital adrenal hyperplasia (CAH); the value and benefits CRENESSITY brings to patients with CAH, including its potential to enable patients to transition toward more physiologic glucocorticoid dosing; the clinical utility, adoption and impact of the newly published expert recommendations and algorithms for glucocorticoid dose reduction in pediatric and adult patients treated with CRENESSITY; the ability of Neurocrine Biosciences to ensure patients have access to CRENESSITY; and whether the results from our clinical trials of CRENESSITY are indicative of real-world results. Factors that could cause actual results to differ materially from those stated or implied in the forward-looking statements include, but are not limited to, the following: risks and uncertainties associated with Neurocrine Biosciences’ business and finances in general, as well as risks and uncertainties associated with the commercialization of CRENESSITY, including the extent to which patients and physicians accept and adopt CRENESSITY; whether CRENESSITY receives adequate reimbursement from third-party payors; risks and uncertainties relating to competitive products and technological changes that may limit demand for CRENESSITY; risks associated with the Company’s dependence on third parties for development and manufacturing activities related to CRENESSITY, and the ability of the Company to manage these third parties; risks that additional regulatory submissions for CRENESSITY may not occur or be submitted in a timely manner; risks that the FDA or other regulatory authorities may make adverse decisions regarding CRENESSITY; risks that post-approval CRENESSITY commitments or requirements may be delayed; risks that CRENESSITY may be precluded from commercialization by the proprietary or regulatory rights of third parties, or have unintended side effects, adverse reactions or incidents of misuse; risks and uncertainties relating to competitive products and technological changes that may limit demand for CRENESSITY; and other risks described in the Company’s periodic reports filed with the Securities and Exchange Commission, including without limitation the Company’s quarterly report on Form 10-Q for the quarter ended March 31, 2026. Neurocrine Biosciences disclaims any obligation to update the statements contained in this press release after the date hereof other than required by law.
© 2026 Neurocrine Biosciences, Inc. All Rights Reserved. CAP-CFT-US-0047 05/2026
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/neurocrine-biosciences-announces-publication-of-expert-recommendations-for-glucocorticoid-dose-reduction-after-initiating-crenessity-crinecerfont-for-the-treatment-of-classic-congenital-adrenal-hyperplasia-302763404.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/neurocrine-biosciences-announces-publication-of-expert-recommendations-for-glucocorticoid-dose-reduction-after-initiating-crenessity-crinecerfont-for-the-treatment-of-classic-congenital-adrenal-hyperplasia-302763404.html
SOURCE Neurocrine Biosciences, Inc.